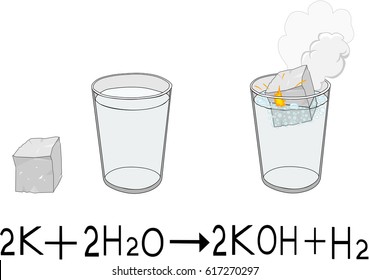
Bases are most often seen in our cleaning cupboards. Bases are substances that are close to 14 on the pH scale. This includes things such as vinegar and lemon juice. Acids are substances that are close to 0 on the pH scale. Acid-Base ReactionsĪn acid–base reaction is a special type of double replacement reaction that occurs between an acid and a base. The most well-known and widely used double replacement reactions are acid-base reactions. Therefore only A can replace C and only B can replace D. In this case A and C are cations and B and D are anions. In it, reactant B replaces reactant D and reactant A replaces reactant C. 
The general formula for a double replacement reaction is: Only cations can replace cations and only anions can replace anions. Cations are positive ions (+) and anions are negative ions (-). In a double replacement reaction, two reactant compounds form two new product compounds through the exchange of ions. Now that you know what happens in a single replacement reaction, what do you think happens in a double replacement reaction? If you thought that two parts switched then you would be right! Heat from the reaction of potassium and water causes the hydrogen gas to combust (see more about combustion reactions below). Another reaction happens soon after the hydrogen gas is produced. The reaction between potassium and water also releases energy in the form of heat. The reaction also releases energy as heat. In it, two molecules of solid potassium (2K) react with two molecules of liquid water (2H2O) to form two molecules of potassium hydroxide (2KOH) in solution and one molecule of hydrogen gas (H2). This oxygen is critical for life on Earth! It allows plants and some microbes to convert water and carbon dioxide gas into storable sugar and oxygen. Photosynthesis is one of the most important chemical reactions on Earth. Some synthesis reactions can result in more than one product. Not all synthesis reactions result in the combination of two substances into one. But when combined, their product is a substance that can put out fires! Hydrogen and oxygen gases are both highly combustible gases. This is a pretty incredible reaction when you think about it. Molecules of water have two of the white shapes stuck to one of the red shapes. Molecules of oxygen gas have two of the red shapes stuck together. Molecules of hydrogen gas have two of the white shapes stuck together. Hydrogen is represented by white, somewhat spherical shapes and oxygen is represented by red, somewhat spherical shapes. Shown are 3D renderings of hydrogen gas, oxygen gas and water molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
